RYDAPT dosage and administration for AML
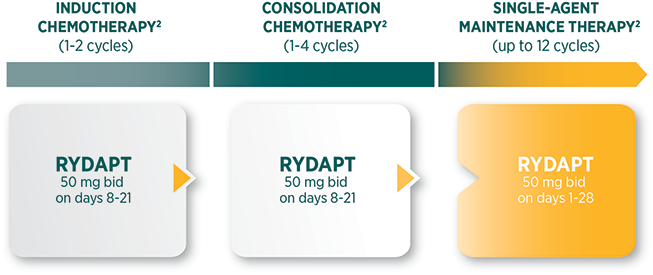
Recommended dosing regimen throughout the course of treatment1
 |
The recommended dose of RYDAPT is 50 mg orally twice daily |
 |
RYDAPT should be taken orally twice daily at approximately 12-hour intervals |
 |
RYDAPT is dosed on days 8 to 21 of induction and consolidation chemotherapy cycles, and then for patients in complete response every day as single-agent maintenance therapy until relapse for up to 12 cycles of 28 days each |
 |
RYDAPT capsules should be swallowed whole with a glass of water |
 |
RYDAPT should be taken with food |
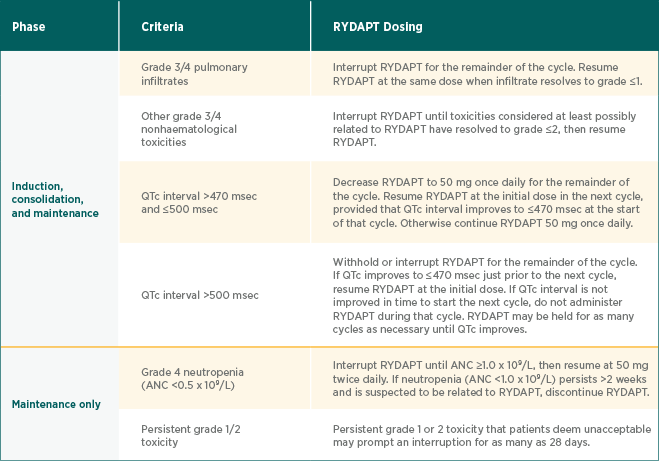
RYDAPT dose interruption, reduction, and discontinuation recommendations in patients with AML1
| Phase | Criteria | RYDAPT Dosing |
| Induction, consolidation, and maintenance | ||
| Grade 3/4 pulmonary infiltrates | Interrupt RYDAPT for the remainder of the cycle. Resume RYDAPT at the same dose when infiltrate resolves to grade ≤1. | |
| Other grade 3/4 nonhaematological toxicities | Interrupt RYDAPT until toxicities considered at least possibly related to RYDAPT have resolved to grade ≤2, then resume RYDAPT. | |
|
QTc interval >470 msec and ≤500 msec |
Decrease RYDAPT to 50 mg once daily for the remainder of the cycle. Resume RYDAPT at the initial dose in the next cycle, provided that QTc interval improves to ≤470 msec at the start of that cycle. Otherwise continue RYDAPT 50 mg once daily. | |
| QTc interval >500 msec | Withhold or interrupt RYDAPT for the remainder of the cycle. If QTc improves to ≤470 msec just prior to the next cycle, resume RYDAPT at the initial dose. If QTc interval is not improved in time to start the next cycle, do not administer RYDAPT during that cycle. RYDAPT may be held for as many cycles as necessary until QTc improves. | |
| Maintenance only | ||
| Grade 4 neutropenia (ANC <0.5 x 109/L) |
Interrupt RYDAPT until ANC ≥1.0 x 109/L, then resume at 50 mg twice daily. If neutropenia (ANC <1.0 x 109/L) persists >2 weeks and is suspected to be related to RYDAPT, discontinue RYDAPT. |
|
|
Persistent grade 1/2 toxicity |
Persistent grade 1 or 2 toxicity that patients deem unacceptable may prompt an interruption for as many as 28 days. |

If a dose is missed, the patient should take the next scheduled dose at the scheduled time1
If vomiting occurs, the patient should not take an additional dose of RYDAPT, but should take the next scheduled dose1
RYDAPT capsules should not be opened, crushed, or chewed to ensure proper dosing and avoid the unpleasant taste of the capsule content1
Prophylactic antiemetics should be administered in accordance with local medical practice as per patient tolerance1
Consult the prescribing information for dose modification recommendations
Packaging1
RYDAPT is dispensed in multipacks containing 112 (4 packs of 28) soft capsules. One blister contains 4 soft capsules. Tell your patients to use only as directed.
Special precautions for storage1
This medicinal product does not require any special temperature storage conditions
Store in the original container in order to protect from moisture
Special precautions for disposal1
Any unused medicinal product or waste material should be disposed of in accordance with local requirements

Not actual size.
Special precautions for storage1
This medicinal product does not require any special temperature storage conditions
Store in the original container in order to protect from moisture
Special precautions for disposal1
Any unused medicinal product or waste material should be disposed of in accordance with local requirements
ANC, absolute neutrophil count; bid, twice daily.
References: 1. RYDAPT [Summary of Product Characteristics]. Novartis Pharma AG; 2017. 2. Data on file. Study no. CPKC412A2301. Novartis Pharmaceuticals Corp; 2016.